近十年以来,油脂浸洗鸡尾酒逐渐成为一种流行风潮。调酒师使用这项技术可以制作各种各样的鸡尾酒饮料,例如 Benton 老式培根波旁威士忌(Benton’s Old-Fashioned, a bacon-infused bourbon cocktail)和温和山核桃黄油波旁威士忌(milder pecan-butter-infused bourbon)。本篇博客文章,我们介绍了这种创新的鸡尾酒技术的制作步骤以及如何将其应用于工业中。实际上,许多行业的很多化学过程已经使用了类似的方法。
油脂浸洗鸡尾酒:乙醇释放香料的能力
油脂浸洗的目的是从肉类和蔬菜中提取油脂中的香料。在油脂浸洗过程中,一些香料可溶于乙醇,乙醇具有溶解亲水性和疏水性溶质的能力。例如,乙醇可以将水溶性柠檬汁与山核桃黄油中的脂肪酸混合以产生新的风味。
乙醇具有的溶解极性和非极性溶质的能力使葡萄酒具有浓郁的风味。这也是使用乙醇作为溶剂时可获得多种口味的主要原因。例如,由于乙醇溶解了橡木桶中的溶质,因此使用橡木桶蒸馏威士忌酒和酿造葡萄酒会增加其口味的复杂性。这些溶质不能被水溶解。

可以通过将乙醇等溶剂与油类混合来提取香料和香精。
在家居用品、化妆品、制药和食品工业的工业制造过程中,我们也可以发现油脂浸洗酒的现象和过程,包括回收维生素、分离香精和香料、脱咖啡因的方法以及更多应用。
下面,我们来了解一下油脂浸洗酒的步骤,以及如何将其应用到工业生产过程。
步骤1:准备油脂
脂肪洗涤过程的第一步是融化或溶解肉或植物脂肪。如果存在固体基质,我们还必须从基质中提取脂肪风味。从橡木桶中提取香料就是一个很好的示例,此过程称为浸出。
为了在工业过程中融化脂肪,我们可以使用配有加热罩的混合器。至于浸出提取,可以使用类似于制作威士忌的方法,但是在大多数情况下,我们可以简单地研磨固体以提高提取率。例如,用烤橡木片制作橡木味的伏特加酒。
如果需要,第二步是从熔融脂肪或油中去除固体。调酒师可以使用过滤器来完成这一过程,该过程称为过滤。在工业规模上,我们用滤饼可以去除的带式过滤器代替过滤器。为了避免油固化成脂肪,需要在高温下进行过滤。
Hawthorne 过滤器是用于鸡尾酒制作过程的重要器具。
油经过滤后,调酒师将其倒入振动筛,然后与酒混合。为了扩大此过程,我们将必须将液体和油送入萃取塔。
步骤2:液-液萃取
在香味的提取过程中,存在一个两相系统:能够溶解疏水性香味的油(熔融脂肪)相和包含疏水性和亲水性风味的乙醇相。现在,我们必须将香味从油相转移到乙醇相,这一过程称为液-液萃取。在液-液萃取过程中,我们需要使每单位体积的相边界最大。这一目标可以通过在乙醇中产生尽可能小的油滴来实现,因此在提取过程中进行混合非常重要。
调酒师只需摇晃油和乙醇的混合物。该过程中会产生大量的液滴,并增强每个相中溶质的传输。

为了对油中的香味进行有效的液-液萃取,我们需要在乙醇溶液中产生小油滴。图片由 JD 提供。在 CC BY 2.0 下获得许可,通过Flickr Creative Commons共享。
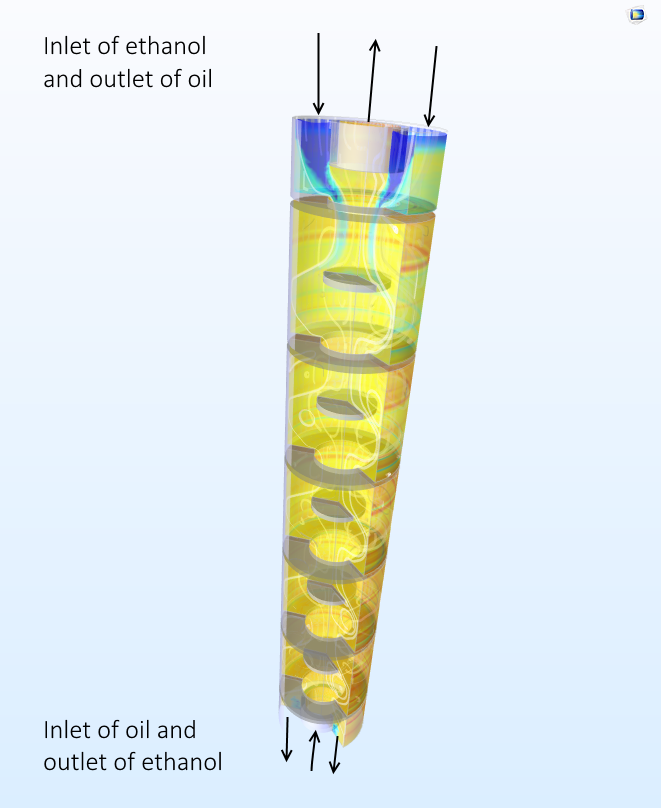
在工业生产过程中,由于使用的容器体积较大,我们可以使油和乙醇流过液-液萃取塔(例如脉冲塔)来代替振动。调酒过程中会扩大的一个复杂因素是温度。在工业萃取塔中,一些油会固化成脂肪。这意味着萃取塔必须保持足够高的温度,以防止油凝固。
为了将油滴分解成较小的油滴,我们需要施加一个超过将每个油滴维持在一起的表面张力的力。我们可以通过在萃取塔中从顶部到底部均匀振动乙醇相,和从底部到顶部均匀振动油相来实现这一目标。就像调酒师的摇晃动作一样,均匀振动增强了溶质与液滴表面之间的传输。
在油和乙醇的交界处,溶质交换非常快。这个过程是如此之快,以至于通常被认为处于平衡状态。相界面两侧的油相和乙醇相中的溶质浓度取决于分配常数:
其中 P 表示分配系数,c 是溶质的浓度。
如果我们在微观尺度上查看一个液滴,溶质在各相之间的传输速率取决于从油滴的本体到相边界,以及从相边界到乙醇相的迁移。我们需要一个非常大的表面积作为相边界,以获得较大的横截面积,使溶质通量往返于该边界。
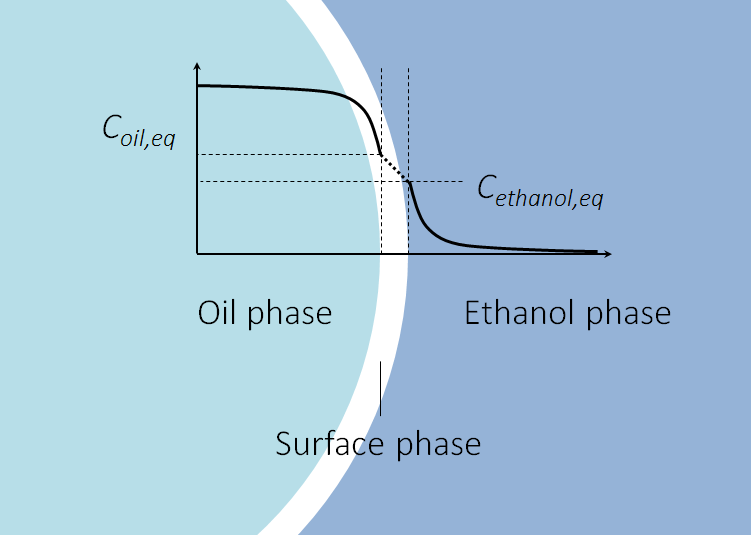
根据吉布斯的研究,存在一个包含油和乙醇分子的混合物的表面相,如下图所示。在该表面相的每一侧,油相和乙醇相中具有平衡的溶质浓度。每个溶质的两个平衡浓度之间的关系由分配系数给出。
当开始提取时,油滴内和液滴周围的乙醇相中的溶质浓度都有很大的梯度,如下图左所示。提取完成后,液滴和乙醇相中各处的浓度根据分配系数等于平衡浓度,如下图右所示。
萃取开始时(左)和萃取完成后(右),乙醇(蓝色)中的油滴(绿松石)。该图显示了沿垂直于相界面并穿过界面沿线的浓度。油相和乙醇相之间被表面相分开,该表面相包含两个相的分子。
下面的动画显示了一个油滴在上升的过程中,将溶质转移到连续相(本例为乙醇)中。当气泡上升时,沿路径会留下一丝溶质。顶部的静置油中也含有溶质,但是运输过程非常缓慢直到液滴碰到表面,这会在相边界处发生一些混合。
在乙醇中上升的油滴。随着油滴的移动,溶质从液滴中提取到连续的乙醇相。在乙醇的顶部还有一层油。
研究一个液滴(例如上方的上升油滴)可以使我们更好地了解微观尺度下发生的过程。但是,即使是制作鸡尾酒,我们也要处理数千滴油滴。在这种情况下,我们需要使用分散两相流模型研究液-液萃取。
在这些模型中,我们不处理油和乙醇之间界面的确切形状,而是将体积分数、气泡大小以及两相之间的特定界面面积作为变量。根据相之间的速度差,每个相内的速度梯度、体积分数、液滴数和尺寸分布,我们可以估算出油相和乙醇相之间的传质系数。这些类型的分散模型用于工业液-液萃取塔的建模和仿真。
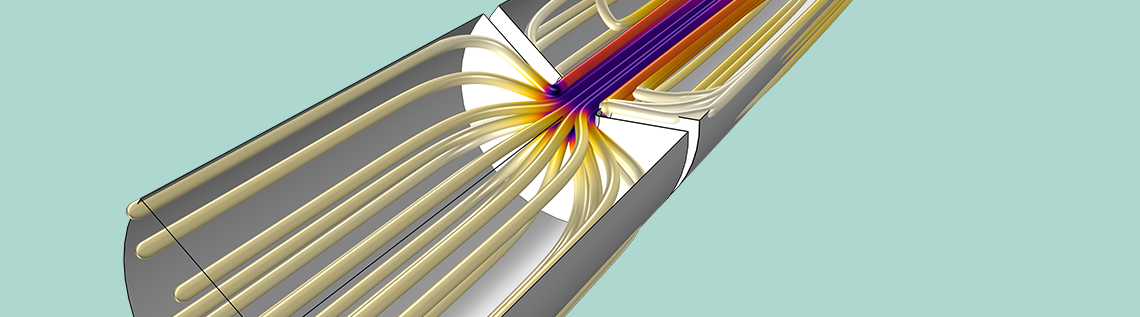
液–液萃取塔的两相流混合模型,显示出油组分在何处是高(红色)和低(蓝色)的。一般而言,塔中的油组分是均衡的,在油相入口和出口的中心聚集。流动是逆向的,乙醇向下流动,而油向上流动。油和乙醇之间有足够的时间来提取香料。
当两个相中溶解的物质的分配可能处于平衡状态时,液-液萃取过程结束。这并不意味着香料的浓度在两相中相等,因为疏水性物质在油相中的浓度始终较高,而亲水性物质在乙醇相中的浓度较高。
萃取完成后,必须让两相系统静置,以使油相作为连续相(而不是液滴在乙醇中的分散相)分离到乙醇顶部。调酒师可以通过让振动筛休息一会来实现这一目标。
步骤3:清除油脂(或其他脂肪)
在工业塔中,油相在顶部作为液相分离,而乙醇可以在塔的底部分离。乙醇中可能仍包含少量分散的油滴,但我们可以使用沉降池将剩余的油分离出来。
一旦调酒师的振动筛中出现两个连续的分离相,他就可以将两相液体放入冰箱。然后,可以将冷冻的脂肪作为固体饼除去。最后,调酒师用滤茶器除去酒中残留的脂肪,剩下清澈的酒作为制作鸡尾酒的基础。
在工业规模上,仅将两相液体冷冻以除去脂肪是无效的。取而代之的是,我们可以使用沉降池,不断地除去池底的液体,并通过溢流口去除顶部的脂肪。在稳定状态下,沉淀池的乙醇上有一层薄薄的油膜,这对减缓乙醇的蒸发也有积极作用。如果某些油凝固成脂肪,可以用刮板将其轻轻刮掉,而无需搅动油槽。
结语
我必须承认我没有尝试过自己浸洗白酒。我强烈反对将完美的威士忌或波旁威士忌与煎培根或火腿中的脂肪混合。也许我会用加入橡木味的伏特加代替。这实际上听起来很美味,并且感觉更接近威士忌和葡萄酒中已经存在的橡木味。

注入橡木味的生命之水-或威士忌。
再说一次,也许我将会喝一杯威士忌加冰块,一边用雪茄研究卡门涡街。






评论 (3)
业昌 郭
2020-11-16Could you please introduce how to deal with the partition coefficient? Thank you very much
Ying (Grace) Xu
2020-11-20Dear 业昌
When using the two-phase flow moving mesh interface, then the partition coefficient is used in the boundary conditions. For the concentration for a solute in one phase, e.g. phase 1, becomes a function of the concentration of the same solute in phase 2:
c1 = f(c2)
For the equation of the solute in phase 2, you can instead use the fact that the flux has to be continuous over the phase boundary:
flux(c1)*n1 = – flux(c2)*n2, where n1 and n2 are the normal outwards vector at the boundary for the respective phase. The principle is demonstrated for a membrane in this model, see specifically slide 5 in the PowerPoint file:
https://www.comsol.com/model/separation-through-dialysis-258
For a dispersed flow model, this becomes more complicated to formulate but on the other hand it is also easier to solve. You then need to have an expression for the mass transfer coefficient at the phase boundary, usually only in the phase where transport is rate determining. This may also yield an additional concentration variable at the phase boundary for each phase and each solute. Then you use the same principle as above but for the homogenized reaction term instead, i.e. for one solute equation you use the equilibrium condition at the boundary and for the other you use the mass transport coefficient to get an equal reaction but with opposite sign in each point in space for the two phases. We will create a few example models with this feature exemplified during the year.
Best regards,
Ed
Zhiyong Guo
2022-07-07Excuse me, do you have this model?