铜集流体溶解
Application ID: 126981
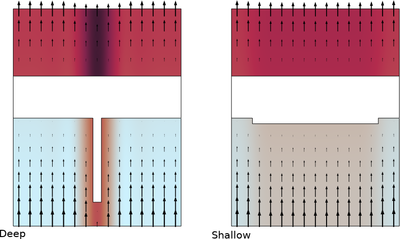
在锂离子电池中,负极石墨电极上的铜集流体在过放电时可能会发生溶解。这不仅会不可逆地损坏集流体,还可能导致铜离子重新沉积并形成枝晶,从而引发安全隐患。为了避免过放电,电池通常会在指定的电压范围内运行,无论是单个电池单元还是整个电池组。然而,过放电现象仍可能发生,例如由于电池组内各单元之间的电荷不平衡,或单个电池单元内部电极的局部不均匀性。本教程将部分探讨后者的影响。
本教程研究了在石墨电极材料涂层厚度不均匀的情况下,过放电过程中铜集流体的溶解现象,并预测了电池单元内部亚铜离子浓度的分布,以及铜重新沉积时的分布情况。
案例中展示的此类问题通常可通过以下产品建模:
您可能需要以下相关模块才能创建并运行这个模型,包括:
建模所需的 COMSOL® 产品组合取决于多种因素,包括边界条件、材料属性、物理场接口及零件库,等等。不同模块可能具有相同的特定功能,详情可以查阅技术规格表,推荐您通过免费的试用许可证来确定满足您的建模需求的正确产品组合。如有任何疑问,欢迎咨询 COMSOL 销售和技术支持团队,我们会为您提供满意的答复。



